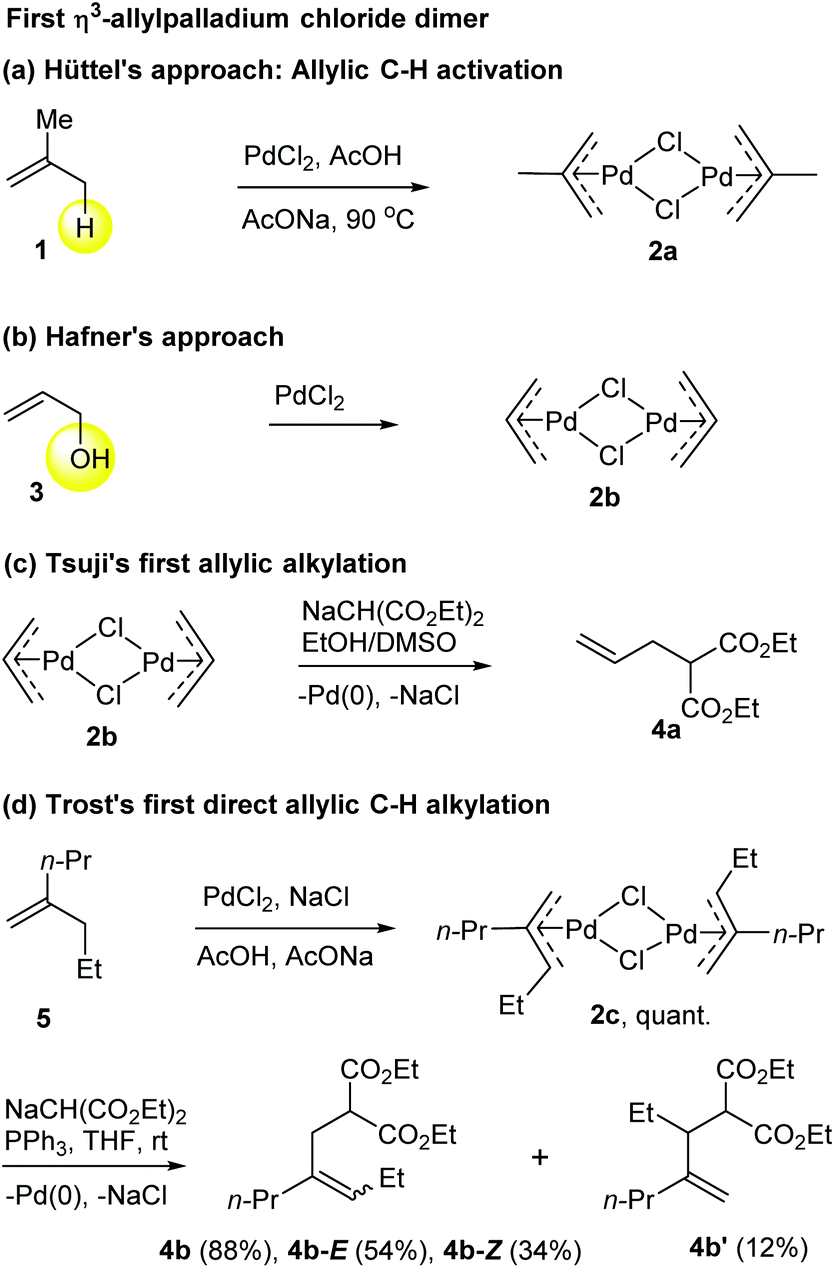
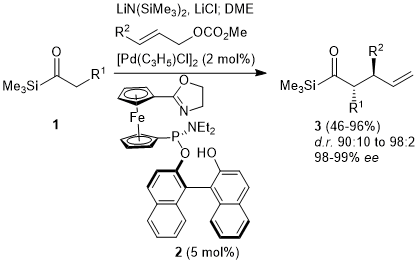
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01725A

Palladium-catalyzed reaction of γ-silylated allyl acetates proceeding through 1,2-shift of a substituent on silicon - ScienceDirect

Influences on the Regioselectivity of Palladium‐Catalyzed Allylic Alkylations - Kazmaier - 2008 - Chemistry – A European Journal - Wiley Online Library

π-Allyl)palladium Complexes Bearing Diphosphinidenecyclobutene Ligands (DPCB): Highly Active Catalysts for Direct Conversion of Allylic Alcohols | Journal of the American Chemical Society

Scope and Mechanism of Allylic C−H Amination of Terminal Alkenes by the Palladium/PhI(OPiv)2 Catalyst System: Insights into the Effect of Naphthoquinone | Journal of the American Chemical Society

Palladium‐Catalyzed anti‐Markovnikov Oxidation of Terminal Alkenes - Dong - 2015 - Angewandte Chemie International Edition - Wiley Online Library

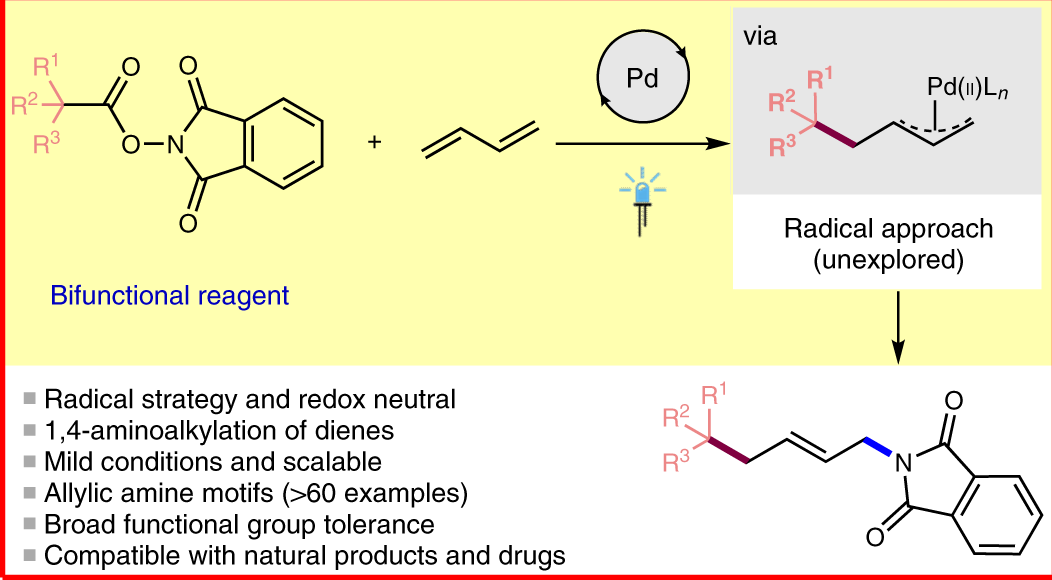

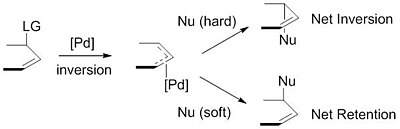
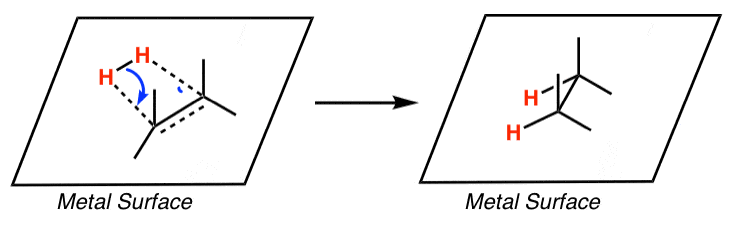
![PDF] Palladium-catalysed alkene chain-running isomerization. | Semantic Scholar PDF] Palladium-catalysed alkene chain-running isomerization. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b5fcd502fc0e60308c690f80805ef2defd55c061/3-Table3-1.png)



![PDF] Palladium-catalysed alkene chain-running isomerization. | Semantic Scholar PDF] Palladium-catalysed alkene chain-running isomerization. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b5fcd502fc0e60308c690f80805ef2defd55c061/2-Table1-1.png)




